Kit for the determination of the CTG triplet repeats number of the DMPK gene by fluorescent fragment analysis
Information about the product
Myotonic dystrophy type 1 or Steinert’s disease is currently the most common form of muscular dystrophy in adults. Inheritance of this multisystem disease is autosomal dominant, and phenotypic expression is highly variable due to an unstable expansion CTG trinucleotide repeats dystrophia myotonica protein kinase gene (DMPK, MIM*605377).
There is a correlation between the number of CTG repeats and the age of appearance and severity of symptoms:
- Mild phenotype (50-100 CTG repeats): associated with clinical manifestations such as mild myotonia or cataracts. The age of appearance for symptoms ranges from 20 to 70 years old.
- Classic phenotype (100-1000 CTG repeats): patients with this phenotype may present myotonia, baldness, cataracts, cardiac arrhythmias, etc. Symptoms appear between 18 and 30 years old.
- Congenital phenotype (>1000 CTG repeats): associated with muscle hypotonia, respiratory distress, intellectual disability, etc. The age of appearance for symptoms ranges from 0 to 18 years old.
Intended use
Adellgene® Myotonic Dystrophy Screening is a semi-automated in vitro diagnostic kit designed for use in clinical laboratories which quantitatively determines the number of repetitions of CTG (cytosine-thymine-guanine) of 3´UTR region of the DMPK gene located in chromosome 19 resulting in Myotonic Dystrophy Type 1 (DM1) disease. It aims to aid diagnosis associated with clinical findings in DM1 that span from mild to severe symptoms.
The use of this kit is the determination of healthy alleles who have between 5 to 49, patients with mild phenotype (50-100 repeats) and classic (100-1000) until 150 repeats. In the cases of homozygous individuals which allele detected is less than 150 repeats or no allele is detected a subsequent study with Adellgene® Myotonic Dystrophy Confirmatory kit must be done to establish the definition of alleles of more than 150 repeats.
The technology is based on the polymerase chain reaction (PCR) of genomic DNA extracted from peripheral blood followed by fluorescence analysis of the size of the PCR fragments obtained by genetic analyzer and conversion of that size in the number of CTG repeats.
Patients who can benefit from this determination are those referred by a specialist. The intended user of the kit is technical personnel trained to carry out the protocol and the interpretation of results described in the instructions for use.
Workflow

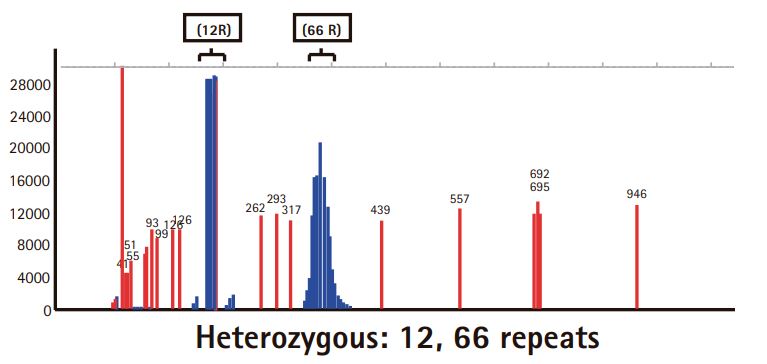
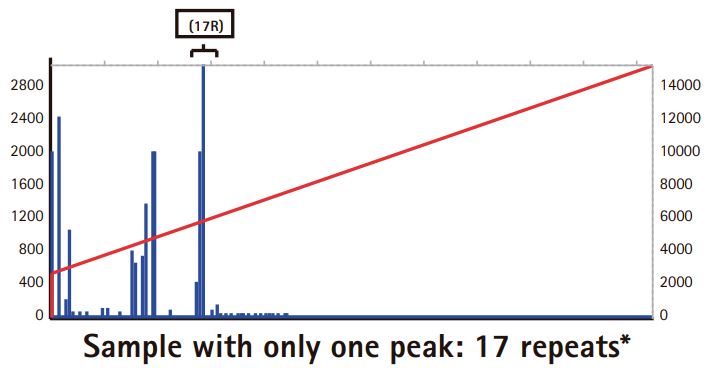
Results
Limitations
- Mutations (point mutations, insertions, deletions) at amplification primer sites are possible and may result in the lack of allele definition. Other technologies could be necessary to resolve the genotyping.
- Homozygous results or when a second allele is not detected, must be confirmed by alternative procedures.
- Data and result interpretation should be revised by qualified personnel.